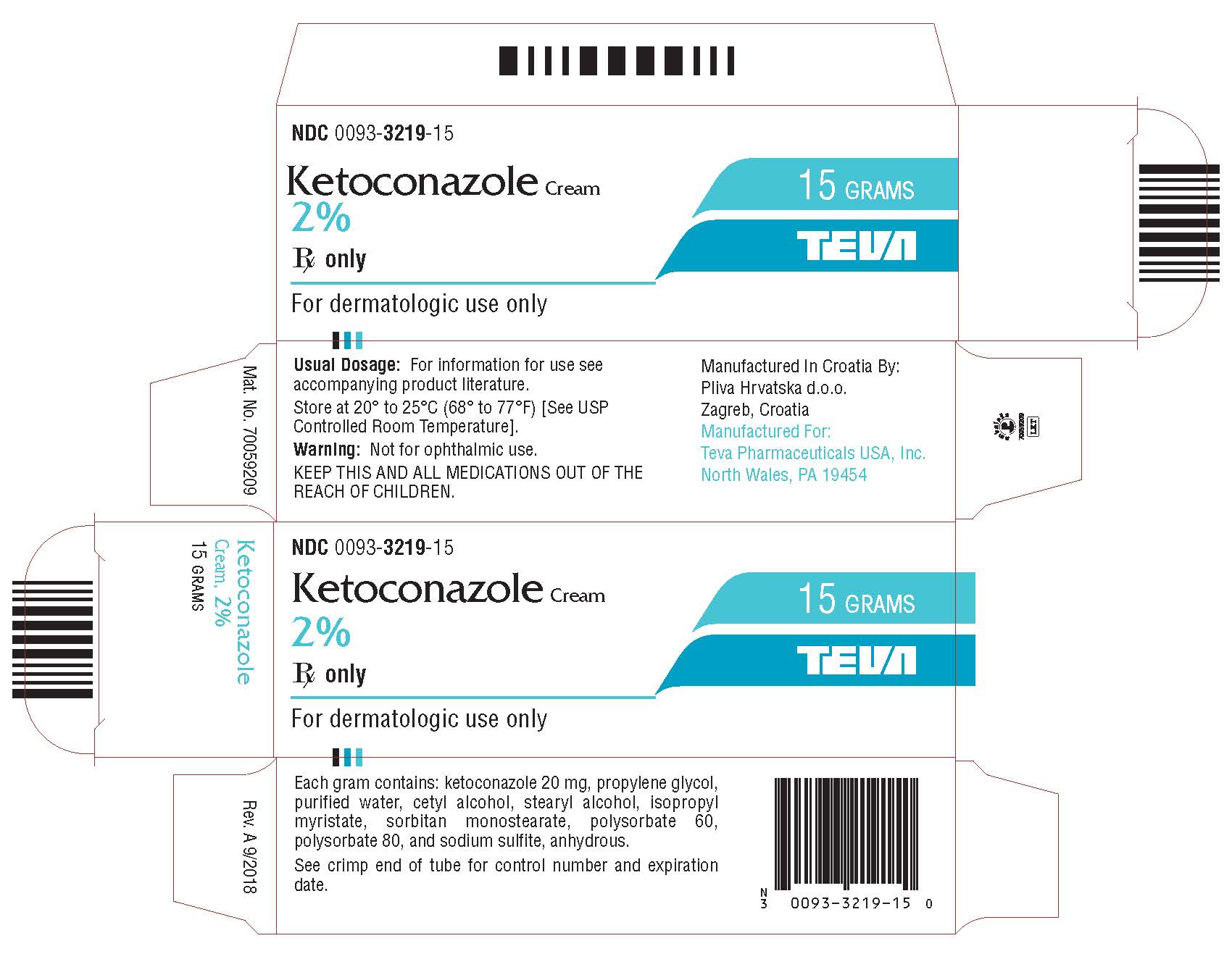
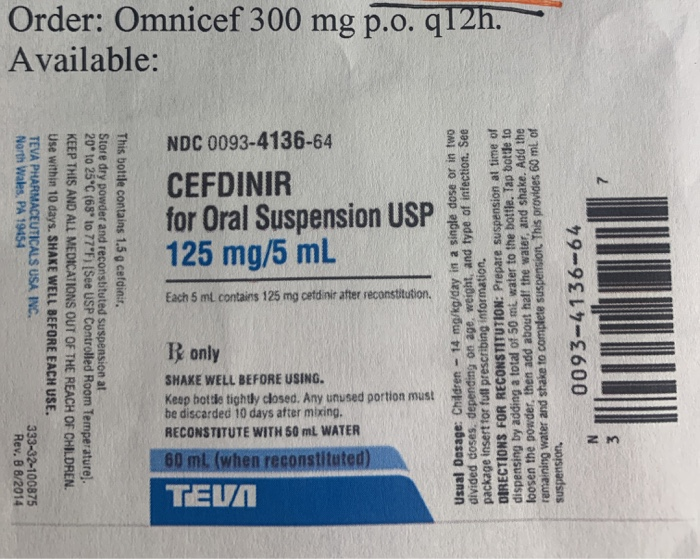
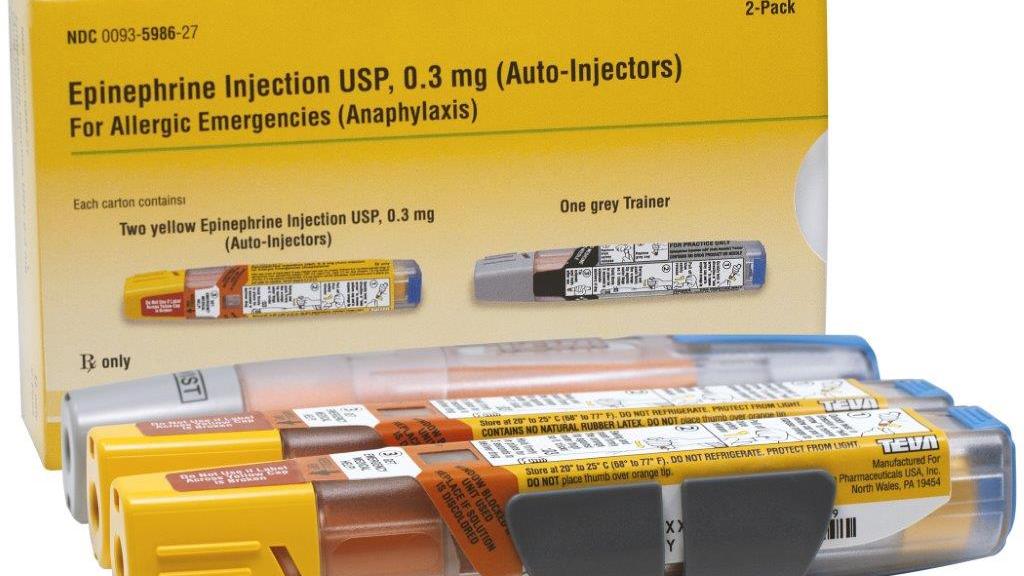
Abrasion-Resistant and Award-Winning: TEVA Relies on Schreiner MediPharm for Novel Adrenaline Injector Label | Packaging Connections
ANDA 077157 TEVA Pharmaceuticals USA Attention: Philip Erickson, R.Ph. Senior Director, Regulatory Affairs 1090 Horsham Road P.O

Teva selects North Jersey site for its new North American headquarters. - Philadelphia Business Journal